Privacy Policy.
Everyone deserves privacy and at PeopleWith we treat privacy and information security in the highest regard.
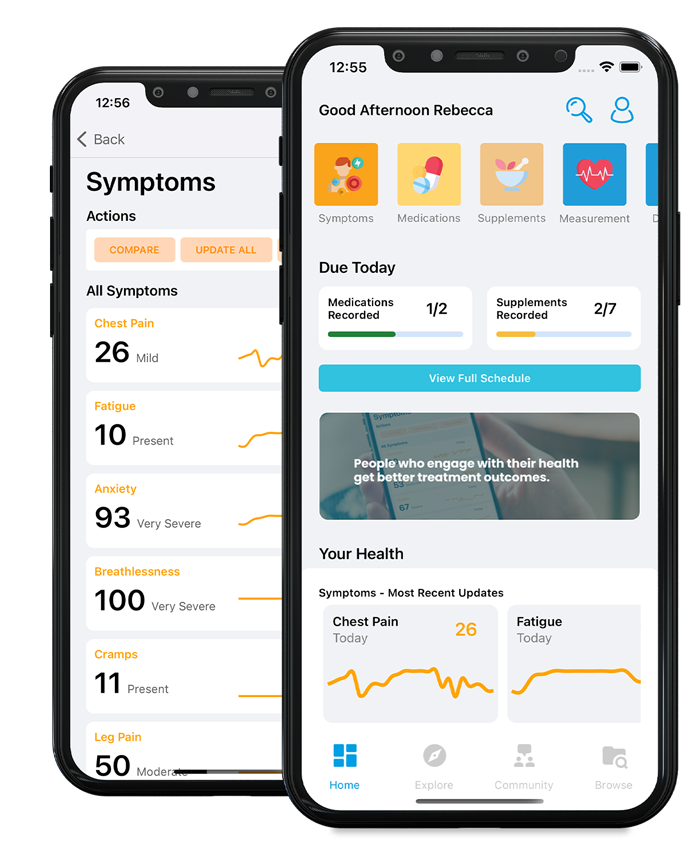
Have you ever wondered why you occasionally get diarrhoea or suffer from vomiting? Well, that’s the kind of thing we spend a lot of time thinking about.
That’s why we’re pleased to say that your surgery is taking part in IID3, a big national study about the gut health. And we’d love it if you would help us by getting involved - whether you suffer from diarrhoea or vomiting a lot, sometimes or never.
This document tells you about why we are doing the study and what we’ll ask you to do if you decide to join in, including consent, data and information requirements, and duration.
This is a large survey. If you decide to join in, you’ll be one of thousands of people from England, Northern Ireland, Scotland and Wales helping us find out more about diarrhoea and vomiting.
Newcastle University is organising and sponsoring IID3. We also have several other partners who will be working with us and GPs all over the UK. The research partners are the University of Oxford (leading on the organisation of the research work with GPs and secure storage of research data), Liverpool Clinical Laboratories and the University of Liverpool (leading on clinical microbiology studies and secure sample storage respectively), the UK Health Security Agency and Public Health Wales (leading on specialist microbiology studies), and Public Health Scotland (leading on surveillance studies). As well as organising the whole study, Newcastle University is also leading on the data analysis.
The research is being funded by the Food Standards Agency, the Government body that makes sure our food safe.
Who has checked this study? Before a study like this can go ahead it undergoes strict checks by an NHS Ethics Committee. This Study has been checked by the East Midlands – Nottingham Research Ethics Committee.
We will find out how many people have diarrhoea or vomiting during a year. We will ask how many of these people go to their doctor when they have diarrhoea. We will also study what germs are causing the symptoms.
Other names for diarrhoea and vomiting are “infectious intestinal disease”, “food poisoning”, “gastroenteritis” and “gastric flu”.
Although there are some official figures about diarrhoea and vomiting, we want to find out how good they are.
You’ll be helping the Food Standards Agency decide whether current food safety measures have worked or if they need to make changes to safety policy. So, it’s an important study that could benefit everyone in the UK.
We do not think that there are any risks in taking part in this study.
If you do agree to take part your GP practice will set you up as a study participant and arrange for you to get regular study updates. All you have to do is:
1. Fill in a consent form completed on the PeopleWith Research Application. This lets us know that you are happy to take part in IID3. Your GP practice will record consent in your medical record.
2. Fill in a short questionnaire which will help us to understand a bit more about you and your illness.
3. Give us a faeces (poo) sample. If you have forgotten to send us a consent form, or initially declined to take part, but you send us a sample anyway, we will assume that we can test the sample that you have provided. We will send the results to your GP. We will then destroy the sample straightaway after we have tested it.
PeopleWith Limited is a company incorporated in Northern Ireland (the Company).
The following terms and conditions form part of the agreement between you the “User” of the PeopleWith application (“Application”) and the Company. This agreement also summarises how you may use the Application and you how the Company may collect, receive, protect and utilise your information.
The Application is available to download via the Apple App Store and the Google Play Store. After downloading the Application, you will be required to create a user account which is specific to the Application. You will be sent confirmation of the registration process by email.
By agreeing to be bound by these Terms and Condition the Company gives you consent to use the Application. If you do not agree to these Terms and Conditions, in whole or in part you may not use or access any part of the Application or the PeopleWith platform and supporting materials.
We may amend this Policy at any time. Any changes we may make will be posted on this page, please check back frequently. Your continued use of the services after posting will constitute your acceptance of, and agreement to, any changes. You will be notified by email and/or application notification.
PeopleWith Limited is registered with the Information Commissioners Office (ICO), with registration reference: ZA745747
If you have any questions about this Policy, please contact info@peoplewith.com. We will endeavour to respond to all requests within 2 working days.
By agreeing to these Terms and Conditions, the Company hereby grants you a non-exclusive, non-transferable right to use the Application, solely for your own personal purposes, subject to the terms and conditions of this Agreement. All rights not expressly granted to you are reserved by the Company.
You may not access the Application if you are a direct competitor of the Company, except with the Company’s prior written consent. In addition, you may not access the Application for purposes of monitoring its availability, performance or functionality, or for any other benchmarking or competitive purposes.
You shall not (i) license, sublicense, sell, resell, transfer, assign, distribute or otherwise commercially exploit or make available to any third party the Application or the content in any way; (ii) modify or make derivative works based upon the Application or the content; (iii) create Internet 'links' to the Application or 'frame' or 'mirror' any Application content on any other server or wireless or Internet-based device; or (iv) reverse engineer or access the Application in order to (a) build a competitive product or service, (b) build a product using similar ideas, features, functions or graphics of the Application, or (c) copy any ideas, features, functions or graphics of the Application.
You may use the Application only for your own personal purposes and shall not: (i) send spam or otherwise duplicative or unsolicited messages in violation of applicable laws; (ii) send or store infringing, obscene, threatening, libellous, or otherwise unlawful material, including material or in violation of third party privacy rights; (iii) send or store material containing software viruses, worms, Trojan horses or other harmful computer code, files, scripts, agents or programs; (iv) interfere with or disrupt the integrity or performance of the Application or the data contained there in; or (v) attempt to gain unauthorised access to the Service or its related systems or networks.
You are responsible for all activity occurring under your User account and shall abide by all applicable local, state, national and foreign laws, treaties and regulations in connection with your use of the Application, including those related to data privacy, international communications and the transmission of technical or personal data. You shall: (i) notify Company immediately of any unauthorised use of any password or account or any other known or suspected breach of security; (ii) report to Company immediately and use reasonable efforts to stop immediately any copying or distribution of Content that is known or suspected by you; and (iii) not impersonate another User or provide false identity information to gain access to or use the Application.
The Company alone shall own all right, title and interest, including all related Intellectual Property Rights, in and to the Company technology, content and the Application and any suggestions, ideas, enhancement requests, feedback, recommendations or other information provided by you or any other party relating to the Application. This Agreement is not a sale and does not convey to you any rights of ownership in or related to the Application, the or the Intellectual Property Rights owned by the Company. The Company name, the Company logo, and the product names associated with the Application are trademarks of the Company, and no right or license is granted to use them.
The Company respects the privacy and confidentiality of all users who engage with the Application, Organisation or Marketing.
For the purposes of applicable data protection laws, The Company is the Data processor for the purposes described in this policy.
The Company adheres to the seven key principles that underpin Data Protection legislation:
- Lawfulness, fairness and transparency
- Purpose limitation
- Data minimisation
- Accuracy
- Storage limitation
- Integrity and confidentiality (security)
- Accountability
These principles are central to how the Company, manage, store and process data.
The Company strives to ensure that all data that is shared with us or within the Application is treated with respect for personal, and client, privacy and protected in line with all our legal responsibilities and recognised best practice standards and processes.
The Company will only collect the minimum levels of personal data necessary to support operational processes and functionality within the Application and will only use your personal data as described within this policy.
Protecting your privacy important to us. For that reason, any of your personal data will only be collected, stored and processed by the Application in compliance with the General Data Protection Regulation 2016/679 (“GDPR”).
The IID3 study research team will need to use information from you, from your medical records and your GP for this research project.
This information will include your:
1. Patient ID
2. Full name
3. Contact details
4. Ethnic group
5. History of bowel problems
6. Symptoms of IID
7. History of foreign travel
8. Results of the stool sample tests
9: Email Address
We’ll also ask about the job of the main earner in the household so that we can make sure that the study covers the whole of society.
People will use this information to do the research or to check your records to make sure that the research is being done properly.
People who do not need to know who you are will not be able to see your name or contact details. Your data will have a code number instead.
We will keep all information about you safe and secure. We will not share any data with the Food Standards Agency that could identify you.
Once we have finished the study, we will keep some of the data so we can check the results. We will write our reports in a way that no-one can work out that you took part in the study.
You can find out more about how we use your information at www.hra.nhs.uk/information-about-patients/ our leaflet available from www.hra.nhs.uk/patientdataandresearch by asking one of the research team by sending an email to data.protection@newcastle.ac.uk
You may give the information about yourself by when you register for an account in respect of the Application for the IID3 Study.
The information you give the Company may include:
- Email Address (Required)
- Password (Required)
- Gender (Required)
- Age/Date of Birth (or age-range) (Required)
The minimum level of data required to register for the service includes, email address, password, date of birth and gender. We need your date of birth to ensure you are of legal age to use the service in your jurisdiction, and your gender to tailor your experience and access to resources.
Should the purpose of data collection change, you will be informed and opt-in consent re-obtained.
Personally Identifiable Information is never accessed by the Company and is utilised for the purposes of personalisation within the Application. In certain circumstances and dependant on Application functionality, consent is required for you to share your information outside of the Application. Consent will be requested and recorded for information governance, auditing and validation. Consent for the IID3 Study is presented at registration within the PeopleWith Research Application.
Visiting the Application, sending us emails, and completing in-app registration constitutes electronic communications. You consent to receive electronic communications, and you agree that all agreements, notices, disclosures, and other communications we provide to you electronically, via email and on the Application, satisfy any legal requirement that such communication be in writing (See: Marketing Activities).
All information you supply remains anonymous, safe and secure. Users have rights to their data which the Company must respect and comply with to the best of our ability. The Company must ensure individuals can exercise their rights in the following ways in accordance with GDPR:
Right to be informed: Keeping a record of how the Company uses personal data to demonstrate compliance with the need for accountability and transparency.
Right of access: Enabling Users to access their personal data and supplementary information. Allowing individuals to be aware of and verify the lawfulness of the processing activities.
Right to rectification: The Company must rectify or amend the personal data of the individual if requested because it is inaccurate or incomplete. This must be done without delay, and no later than one month.
Right to erasure: Individuals have a right to have their data erased and for processing to cease. The User has the right to delete or remove individual personal data any time. The User can remove their data by deleting their profile in the Application.
Right to restrict processing: The Company must comply with any User request to restrict, block, or otherwise suppress the processing of personal. The Company is permitted to store personal data if it has been restricted, but not process it further.
Right to object: The Company respects the right of an individual to object to data processing based on legitimate interest or the performance of a public interest task. The Company respects the right of an individual to object to direct marketing, including profiling.
You (the User) have the right to request access to your data please place your request in an email to dpo@peoplewith.com quoting “Right of Access” in the email subject line. The Company will endeavour to respond to your request within 2 working days.
We do not knowingly collect Personal Data online from individuals under 18. If you become aware that a child has provided us with Personal Data without parental consent, please contact us at: support@peoplewith.com. If we become aware that an individual under 18 has provided us with Personal Data without parental consent, we will take steps to remove the data and cancel that individual’s account.
You can stop being part of the study at any time, without giving a reason, The information you have given up to that point will still be helpful to us.
If you wish to withdraw (leave) the IID3 Study, you can end your participation by choosing the “Withdraw from study” option within the “Profile” section of the PeopleWith Research Application. Your participation will be terminated immediately.
Alternatively, you can request to withdraw (leave) the study by emailing: support@peoplewith.com with the subject line “Withdraw from IID3 Study”. We aim to respond to all withdrawal requests within 2 working days.
Researchers need to manage your records in specific ways for the research to be reliable. This means that we won’t be able to let you see or change the data we hold about you.
If you agree to take part in this study, you will have the option to take part in future research using your data saved from this study. With your permission your sample would be stored securely for a minimum of five years in the Liverpool University Biobank. If you forget to complete the consent form we will not store your sample.
Data automatically captured by the Company’s Web and Application is anonymised and utilised for the purposes outlined below:
- Usage data from use of the web and application including information about your device, information about your visit and how you use the web or application.
- Analytics including bug reporting and problem identification
- Service improvements and product development.
You (the User) are not able to opt out of automatically shared data because data collected is anonymised and aggregated for service improvements. Individuals data cannot be identified and therefore cannot be removed from aggregated service improvement analytics.
The Company do not utilise or share any information you supply to inform marketing activities by Third Parties. The Company may from time to time communicate with you via email for the purposes outlined below:
- Health related research.
- Notifications on changes to the Terms and Conditions of Use and Data Policy.
- Newsletter containing New Features and Product Development Announcements
- Company Specific Events related to your profile.
- Other products or services supplied by the Company.
You (the User) have the ability to opt out of all marketing activities above, apart from “Notifications on changes to the Terms and Conditions of Use and Data Policy” because changes must be agreed to in order to continue the use of the service.
To opt out, please email: info@peoplewith.com with “Unsubscribe” in the subject line. Please contact us at: support@peoplewith.com if you require any assistance with unsubscribing. The Company will endeavour to respond to your request within 2 working days.
The Company retains the data you the user input into the Application for the term and duration of use of the Application in order to ensure normal Application functionality. Upon termination of Application usage, the Company will retain data for a period of two years unless specifically requested to erase the data under data protection laws.
On the event that you the user decide to delete your Account via the Application, systematic processes will permanently delete all data within forty eight hours. Deletion process cannot be reverse once implemented.
The Company utilise ISO27001 certified cloud computing platform Microsoft Azure as its cloud storage provide. Adequate organisational and technical safeguards are implemented to ensure data security.
Data is encrypted at all stages within the PeopleWith Platform, in-flight and at rest. Data encryption is implemented in the following ways:
Transparent Data Encryption: Data moving through PeopleWith’s Platform, from Azure Storage and Databases to the front end of the User Interface, is encrypted utilizing Azure’s Transparent Data Encryption protocols. When data arrives at is destination, it is unencrypted and presented accordingly.
Database Encryption: Data stored in a rest state in PeopleWith’s databases are encrypted with Database Encryption Keys implemented, dynamically rotated & updated on a period basis to ensure maximum defence against data breach.
Data is encrypted in-flight utilising Organisation Level Domain Verification Secure Socket Layer (SSL) for maximum verification and security to facilitate the processing of data through the internet.
The Company is currently working through Cyber Security Essentials framework and seeking to complete ISO27001 Certification from UK certifier BSI in 2024.
We have put in place appropriate security measures to prevent your personal data from being accidentally lost, used or accessed in an unauthorised way, altered or disclosed. In addition, we limit access to your data to those designated employees and contractors who have a business need to know. They will only process your data on instruction, and they are subject to a duty of confidentiality.
We have put in place procedures to deal with any suspected personal data breach and will notify you and The Information Commissioner’s Office of a breach where we are legally required to do so. Additionally, if you have the right to complain to the Information Commissioner’s Office whose details can be found at: https://ico.org.uk.
IID3 Study researchers will publish the results as a report, in medical journals and presented at conferences. Your name and information that can identify you will not be used. If you would like us to send you a summary of the results, please select the box on the consent form. This will be available from the end of 2026.
We will ask your permission to contact you in the future to find out if you are interested in taking part in related research.
You, the User, are entitled to stop using the Application and to terminate this agreement at any time. To do this you must deactivate your account via the Application. This can be done by access the “Withdraw from Study” option from the “Profile” section.
In the event that you feel that you can no longer agree to the present Terms and Conditions you must stop using the Application immediately.
The Company has the right to terminate this agreement by providing you with two weeks’ notice by email.
Either party’s right to terminate the agreement for cause remain unaffected. Cause that entitles the Company to terminate the agreement may be, in particular but not exclusively, User violation under terms outlined in paragraph 2.
If you the User or Company deactivates your User account or withdraws your right to access the Application, the Company will delete any personal data stored about you in accordance with our Privacy Policy.
If we become aware that an individual under 18 has provided us with Personal Data without parental consent, we will take steps to remove the data and cancel that individual’s account.
The Application is not a medical diagnosis tool, nor does the Company claim that the Application be used as a diagnostic tool. If you have concerns regarding your health or are worried about the progression of symptoms you should seek professional medical advice.
You shall indemnify and hold the Company, its officers, directors, employees, attorneys and agents harmless from and against any and all claims, costs, damages, losses, liabilities and expenses (including attorneys' fees and costs) arising out of or in connection with: (i) a claim alleging that use of the your data infringes the rights of, or has caused harm to, a third party; (ii) a claim, which if true, would constitute a violation by you of your representations and warranties.
The Company shall indemnify and hold you harmless from and against any and all claims, costs, damages, losses, liabilities and expenses (including attorneys' fees and costs) arising out of or in connection with: (i) a claim alleging that the Application directly infringes copyright or trademark of a third party; (ii) a claim, which if true, would constitute a violation by the Company of its representations or warranties.
The Company reserves the right to modify the terms and conditions of this Agreement or its policies relating to the Application at any time, effective upon posting of an updated version of this Agreement on the Web or Application. You are responsible for regularly reviewing this Agreement. Continued use of the Application after any such changes shall constitute your consent to such changes.
Where the purpose of data collection or processing activities change, you will be informed via email and opt-in consent reobtained.
This Agreement shall be governed by and construed in accordance with the laws of England.
If you are not sure about any aspect of this study you should ask to speak to the practice where they will try to answer your questions.
If you are unhappy and wish to complain you can do this through the NHS Complaints Procedure (england.contactus@nhs.net) stating ‘For the attention of the complaints team’ in the subject line. Alternatively you can contact the Research Integrity and Governance Manager at Newcastle University (res.policy@ncl.ac.uk).
During office hours: sponsorship@newcastle.ac.uk
Out of office hours: sponsorship@newcastle.ac.uk
Complaints: sponsorship@newcastle.ac.uk
If you have any questions about this Policy, please contact info@peoplewith.com. We will endeavour to respond to all requests within 2 working days.
The Company can be contacted by post at the registered address:
PeopleWith Limited
Unit D10
Omagh Enterprise Centre,
41 – 43 Great Northern Road,
Omagh, Co. Tyrone
BT78 5LU
Northern Ireland
The Company Data Protection Officer can be contacted at the above address or:
Chris Johnston
Data Protection Officer (DPO)
dpo@peoplewith.com
The Third Infectious Intestinal Disease Study (IID3) and PeopleWith Terms and Conditions of Use, Study Information, Consent, Privacy and Data Policy - January 2024